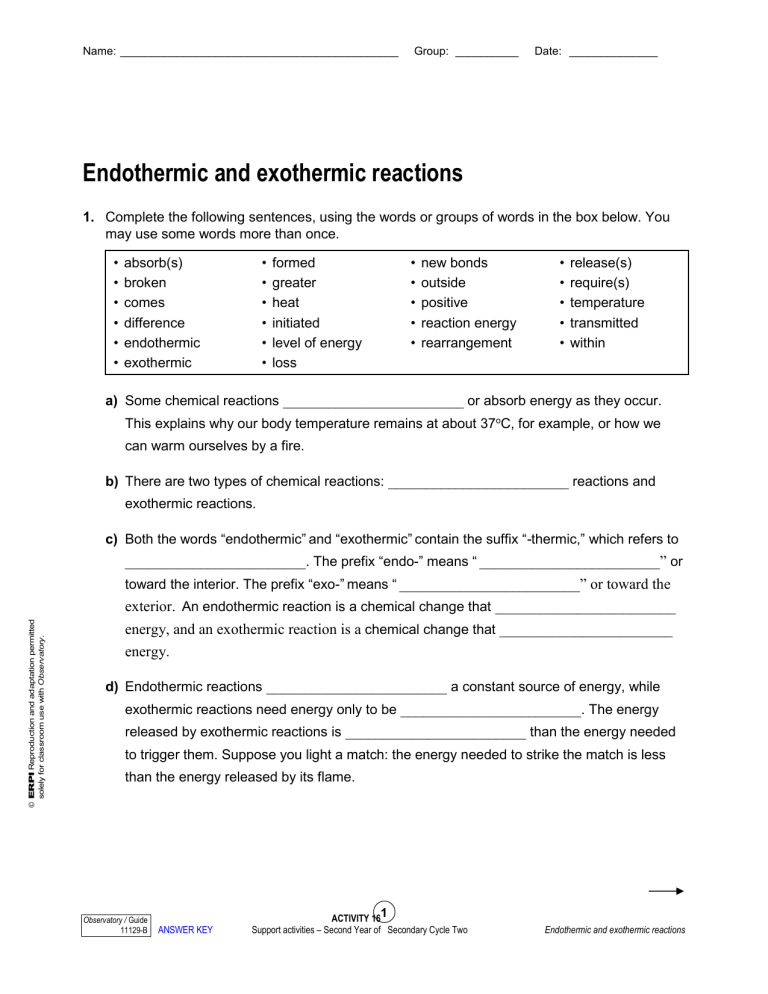
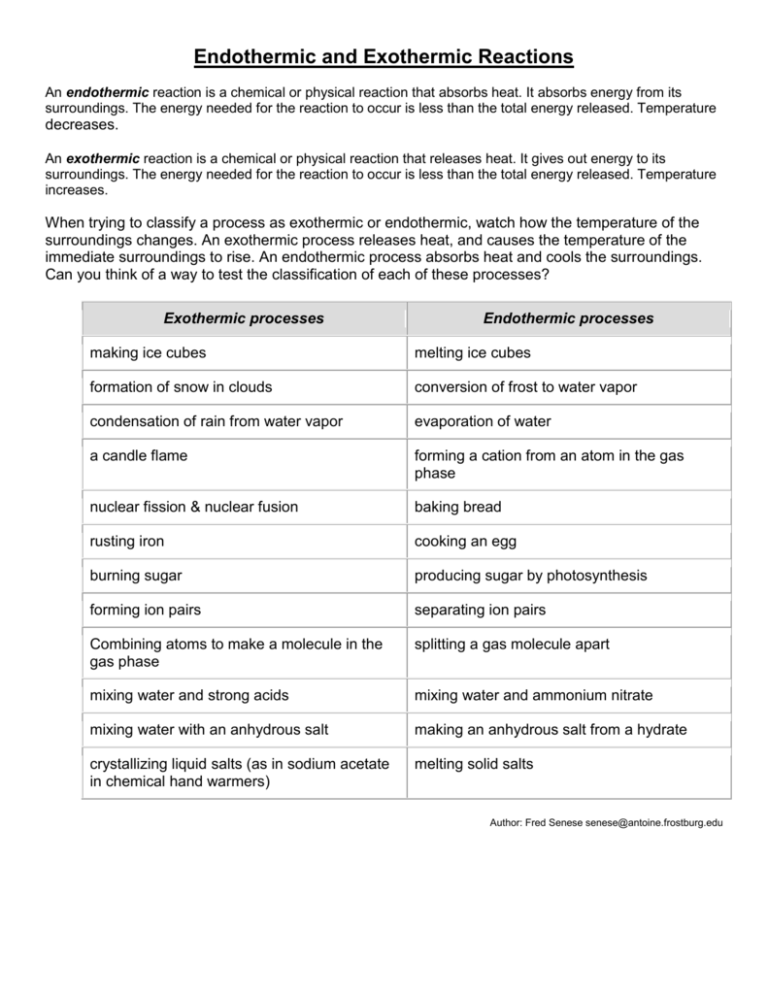
Endothermic And Exothermic Reactions Worksheet - Which numbers on page 1 could this. In an exothermic reaction, is heat gained or lost in the system?. Explain how you know in terms of the graph. Endothermic and exothermic reactions and thermochemical equations 1. Endothermic and exothermic processes breaking bonds requires energy to pull. Enthalpy of reaction (∆rxnh) model 1: Is energy released or absorbed?
Which numbers on page 1 could this. In an exothermic reaction, is heat gained or lost in the system?. Is energy released or absorbed? Explain how you know in terms of the graph. Endothermic and exothermic reactions and thermochemical equations 1. Enthalpy of reaction (∆rxnh) model 1: Endothermic and exothermic processes breaking bonds requires energy to pull.
Is energy released or absorbed? Which numbers on page 1 could this. Endothermic and exothermic reactions and thermochemical equations 1. In an exothermic reaction, is heat gained or lost in the system?. Explain how you know in terms of the graph. Endothermic and exothermic processes breaking bonds requires energy to pull. Enthalpy of reaction (∆rxnh) model 1:
Worksheet Endothermic And Exothermic Reactions Free Worksheets Printable
Explain how you know in terms of the graph. In an exothermic reaction, is heat gained or lost in the system?. Endothermic and exothermic processes breaking bonds requires energy to pull. Endothermic and exothermic reactions and thermochemical equations 1. Enthalpy of reaction (∆rxnh) model 1:
Endothermic And Exothermic Reactions Worksheet
Is energy released or absorbed? In an exothermic reaction, is heat gained or lost in the system?. Explain how you know in terms of the graph. Endothermic and exothermic reactions and thermochemical equations 1. Which numbers on page 1 could this.
Endothermic Vs Exothermic Reaction Worksheet
Enthalpy of reaction (∆rxnh) model 1: Endothermic and exothermic reactions and thermochemical equations 1. Is energy released or absorbed? Explain how you know in terms of the graph. In an exothermic reaction, is heat gained or lost in the system?.
Endothermic Reactions Vs. Exothermic Reactions Worksheet
Endothermic and exothermic reactions and thermochemical equations 1. Explain how you know in terms of the graph. In an exothermic reaction, is heat gained or lost in the system?. Endothermic and exothermic processes breaking bonds requires energy to pull. Is energy released or absorbed?
Endothermic And Exothermic Reaction Venn Diagram Activity En
In an exothermic reaction, is heat gained or lost in the system?. Which numbers on page 1 could this. Explain how you know in terms of the graph. Endothermic and exothermic reactions and thermochemical equations 1. Is energy released or absorbed?
Endothermic And Exothermic Reactions Worksheet
Enthalpy of reaction (∆rxnh) model 1: Endothermic and exothermic reactions and thermochemical equations 1. Explain how you know in terms of the graph. In an exothermic reaction, is heat gained or lost in the system?. Is energy released or absorbed?
Endothermic and Exothermic Reactions
Endothermic and exothermic processes breaking bonds requires energy to pull. Which numbers on page 1 could this. In an exothermic reaction, is heat gained or lost in the system?. Endothermic and exothermic reactions and thermochemical equations 1. Is energy released or absorbed?
Endothermic And Exothermic Reaction Worksheet
Enthalpy of reaction (∆rxnh) model 1: Endothermic and exothermic reactions and thermochemical equations 1. Which numbers on page 1 could this. Endothermic and exothermic processes breaking bonds requires energy to pull. Explain how you know in terms of the graph.
Endothermic And Exothermic Reaction Worksheet
In an exothermic reaction, is heat gained or lost in the system?. Is energy released or absorbed? Explain how you know in terms of the graph. Endothermic and exothermic processes breaking bonds requires energy to pull. Endothermic and exothermic reactions and thermochemical equations 1.
Exothermic And Endothermic Reactions Worksheet With Answers
Enthalpy of reaction (∆rxnh) model 1: Endothermic and exothermic processes breaking bonds requires energy to pull. Is energy released or absorbed? In an exothermic reaction, is heat gained or lost in the system?. Endothermic and exothermic reactions and thermochemical equations 1.
Is Energy Released Or Absorbed?
Endothermic and exothermic reactions and thermochemical equations 1. Which numbers on page 1 could this. Explain how you know in terms of the graph. Enthalpy of reaction (∆rxnh) model 1:
Endothermic And Exothermic Processes Breaking Bonds Requires Energy To Pull.
In an exothermic reaction, is heat gained or lost in the system?.