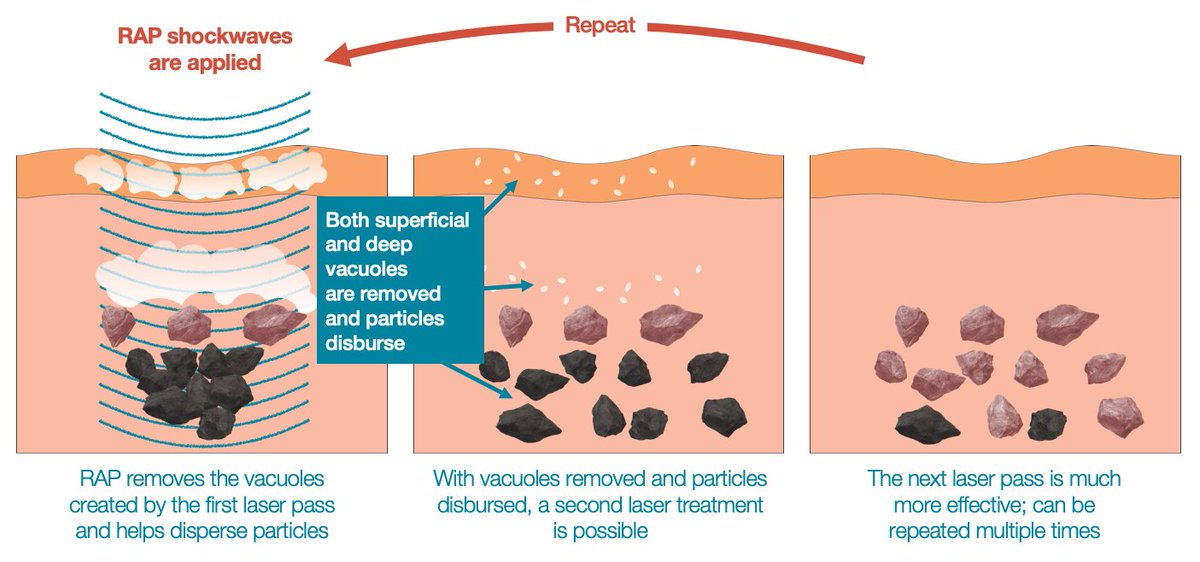
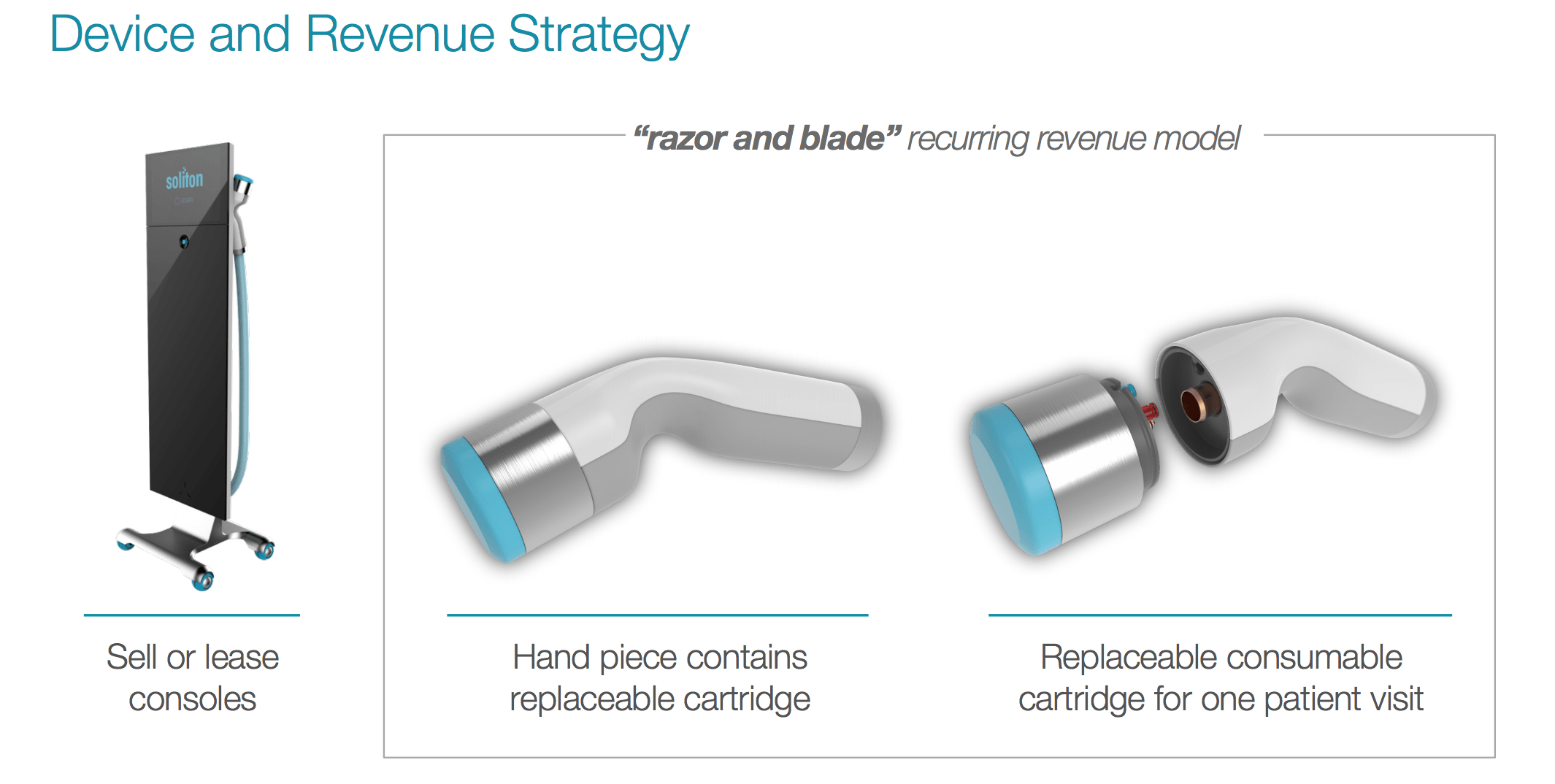
Soliton Tattoo Removal - Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. The technology has received u.s. Food and drug administration granted soliton, inc. 510(k) for modifications to its resonic device. Soliton has received 510 (k) clearance from the u.s.
Soliton has received 510 (k) clearance from the u.s. 510(k) for modifications to its resonic device. Food and drug administration granted soliton, inc. The technology has received u.s. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo.
Food and drug administration granted soliton, inc. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. The technology has received u.s. Soliton has received 510 (k) clearance from the u.s. 510(k) for modifications to its resonic device.
The role of ’biotics in skincare
Soliton has received 510 (k) clearance from the u.s. The technology has received u.s. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. Food and drug administration granted soliton, inc. 510(k) for modifications to its resonic device.
Soliton Tattoo Removal Uk MREBU
Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. Soliton has received 510 (k) clearance from the u.s. 510(k) for modifications to its resonic device. Food and drug administration granted soliton, inc. The technology has received u.s.
Soliton Tattoo Removal Uk MREBU
Soliton has received 510 (k) clearance from the u.s. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. The technology has received u.s. Food and drug administration granted soliton, inc. 510(k) for modifications to its resonic device.
Soliton Soars a Second Day After FDA Approves Tattoo Removal Device
The technology has received u.s. Soliton has received 510 (k) clearance from the u.s. Food and drug administration granted soliton, inc. 510(k) for modifications to its resonic device. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo.
Soliton Will Deliver RESONIC DualPlatform Device June 2021 Plastic
Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. The technology has received u.s. 510(k) for modifications to its resonic device. Food and drug administration granted soliton, inc. Soliton has received 510 (k) clearance from the u.s.
Soliton Faster Tattoo Removal Using RAP Technology Tattoo removal
Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. Food and drug administration granted soliton, inc. Soliton has received 510 (k) clearance from the u.s. 510(k) for modifications to its resonic device. The technology has received u.s.
Soliton Slow, Steady Progress Could Win Tattoo & Cellulite Removal
Soliton has received 510 (k) clearance from the u.s. Food and drug administration granted soliton, inc. 510(k) for modifications to its resonic device. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. The technology has received u.s.
Soliton Acoustic Shockwave Device to Aid in Tattoo Removal Medgadget
Food and drug administration granted soliton, inc. Soliton has received 510 (k) clearance from the u.s. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. 510(k) for modifications to its resonic device. The technology has received u.s.
Acoustically Powered Tattoo Removers Soliton Rapid Acoustic Pulse System
Soliton has received 510 (k) clearance from the u.s. The technology has received u.s. 510(k) for modifications to its resonic device. Food and drug administration granted soliton, inc. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo.
Soliton Will Deliver RESONIC DualPlatform Device June 2021 Plastic
510(k) for modifications to its resonic device. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo. Food and drug administration granted soliton, inc. The technology has received u.s. Soliton has received 510 (k) clearance from the u.s.
The Technology Has Received U.s.
510(k) for modifications to its resonic device. Food and drug administration granted soliton, inc. Soliton has received 510 (k) clearance from the u.s. Fda for its generation ii rapid acoustic pulse (rap) device for tattoo.